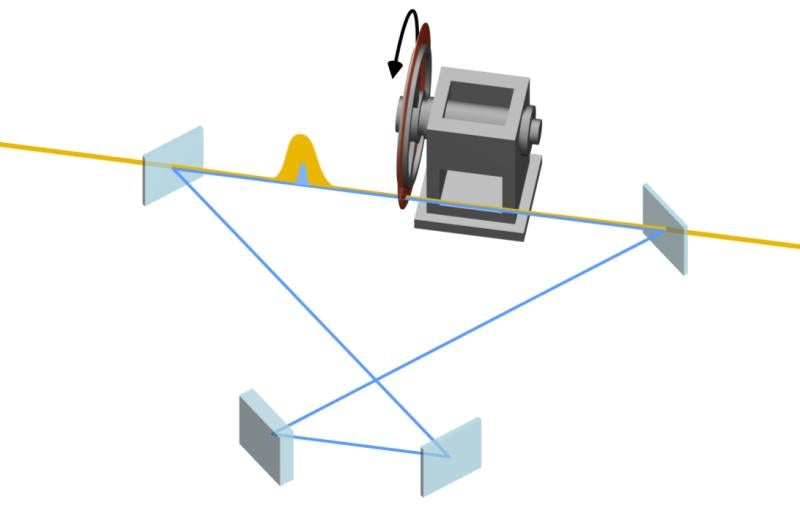
Meet the new instruments that will take advantage of SLAC’s upgraded X-ray laser
Teams at SLAC installed new experimental hutches with cutting-edge instruments that will harness the upgraded facility’s new capabilities and expand the breadth of research done at the facility.
Teams at the U.S. Department of Energy’s SLAC National Accelerator Laboratory recently celebrated the completion of a major upgrade to the world’s first hard X-ray free-electron laser (XFEL), the Linac Coherent Light Source (LCLS) at the US Department of Energy’s SLAC National Accelerator Laboratory. When fully commissioned, its beam will be an average of 10,000 times brighter and produce 8,000 times more pulses per second than the original, up to a million pulses per second, enabling fundamental new insights in materials, physical, chemical and biological sciences.
But enabling extraordinary science takes more than simply shooting powerful laser pulses at things – it also takes cutting-edge scientific instruments and techniques that will harness the upgraded facility’s new capabilities and expand the breadth of research done at the facility. Here's a look at what some of those instruments and techniques can do.
Bouncing electrons
Two of LCLS's new instruments, chemRIXS and qRIXS, implement a method called resonant inelastic X-ray scattering (RIXS). The idea is to scatter X-ray pulses off a material, driving electrons deep inside its atoms into higher energy states. By measuring the light the electrons give off as they settle back down into their original spots, researchers can probe a material's properties in fine detail.

“RIXS is a technique that has developed pretty rapidly,” said Georgi Dakovski, SLAC’s lead scientist for these instruments. “It’s capable of telling us a lot of useful information about how materials behave that isn’t accessible via any other method."
And now, with the LCLS-II upgrade, RIXS can do more than take static pictures, Dakovski said. "We can actually see how materials transform over time. We can follow how energy flows within a material – watching the interplay between electronic charge and spin, and how the atoms vibrate and interact. This energy flow occurs on extremely fast timescales. To “capture” it, one needs a very high flux of femtosecond optical and X-ray pulses. This is where LCLS-II, with its unprecedented increase in average brightness, will provide transformational capabilities."
That ability will come in handy for studying molecules such as photosystem II (PS-II), a key protein complex in plants, algae and cyanobacteria that splits water, producing the oxygen we breathe. Although XFELs have made tremendous progress capturing the intermediate states of that process, scientists are still working to capture the transitional moments between these states and the flow of electric charge throughout the reaction cycle. Experiments at chemRIXS will offer more insight into this process, which can inform the next generation of artificial photosynthetic systems that produce cleaner and more renewable energy from sunlight and water.
At qRIXS, scientists will use the same basic technique to investigate the properties in quantum materials that give rise to room-temperature superconductivity and other phenomena.

Of particular interest are copper oxides, or cuprates, a class of unconventional superconducting materials that can carry electrical current with no loss at unexpectedly high temperatures. Recent research has highlighted the role of charge density waves – static stripes of higher and lower electron density running through a material – and the transition between these waves and other states, although many questions remain. With qRIXS, researchers will be able to study these states in great detail, yielding new insights into the much-debated mechanisms of high-temperature superconductivity.
Watching chemistry happen
The Time-resolved Atomic, Molecular and Optical Science (TMO) instrument, follows in the footsteps of the now-retired AMO instrument, where scientists developed a way to measure ultrashort X-ray pulses that last for just several hundred attoseconds (or billionths of a billionth of a second). The method for producing such short X-ray pulses is called X-ray laser-enhanced attosecond pulse generation (XLEAP), which delivers pulses that are millions of times brighter than the pioneering work that led to the 2023 Nobel Prize in Physics. This approach allows scientists to investigate how electrons zipping around molecules jumpstart key processes in biology, chemistry, materials science and more.

TMO features two locations, or interaction points, for measurements. The first is very versatile, focused on measurement of electrons ejected from an atom when X-rays interact with molecular samples. The spectrum of these electrons provides information on the chemical (bonding) environment near different atoms in the molecule. Ultrashort pulses of soft, or lower-energy, X-rays gives scientists a new way to study transient energy and charge flow – and what functions those enable – in both simple and complex molecular systems. Understanding isolated systems down to atomic lengths and attosecond timescales helps researchers design new catalysts and clean energy sources with reduced impact on the environment – providing a fundamental basis to enhance the sustainability of our society.
The second interaction point in TMO enables the study of isolated quantum systems, making use of a technique called reaction microscopy. Intense X-rays interact with a molecular sample, stripping away electrons and causing the molecule to explode. The reaction microscope collects the exploding fragments and uses this information to reconstruct the structure of the molecular system before the X-ray pulse arrived. This new type of “imaging” provides insights into the motion of molecules that is central to many important processes such as photosynthesis, catalysis and the making and breaking chemical bonds, which is poorly understood on the quantum level.

“Let's say you want to look at a molecule,” said James Cryan, SLAC’s TMO instrument lead scientist. “You can learn a great deal about the electrons in the molecule by measuring what color (or energy) X-rays it absorbs and how that absorption changes with time. Currently, it's challenging for us to get a lot of colors and time points. It can take multiple days, or even multiple beam times, to gather enough meaningful data for basic understanding. The high repetition rate of LCLS-II will allow us to do a full scan of the molecule very quickly. The increased data rate will allow us to measure multiple properties of the molecule and correlate all of them together to gain a better understanding of how it functions.”
Armed with its new capabilities and an updated suite of instruments, LCLS-II will help researchers address a myriad of grand challenges in science by capturing detailed snapshots of rapid processes that are beyond the reach of other light sources.
Contact
For questions or comments, contact the SLAC Office of Communications at communications@slac.stanford.edu.
About SLAC
SLAC National Accelerator Laboratory explores how the universe works at the biggest, smallest and fastest scales and invents powerful tools used by researchers around the globe. As world leaders in ultrafast science and bold explorers of the physics of the universe, we forge new ground in understanding our origins and building a healthier and more sustainable future. Our discovery and innovation help develop new materials and chemical processes and open unprecedented views of the cosmos and life’s most delicate machinery. Building on more than 60 years of visionary research, we help shape the future by advancing areas such as quantum technology, scientific computing and the development of next-generation accelerators.
SLAC is operated by Stanford University for the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time.