Researchers use attosecond X-ray pulses to track electron motion in a highly excited quantum state of matter
Less than a millionth of a billionth of a second long, attosecond X-ray pulses allow researchers to peer deep inside molecules and follow electrons as they zip around and ultimately initiate chemical reactions.
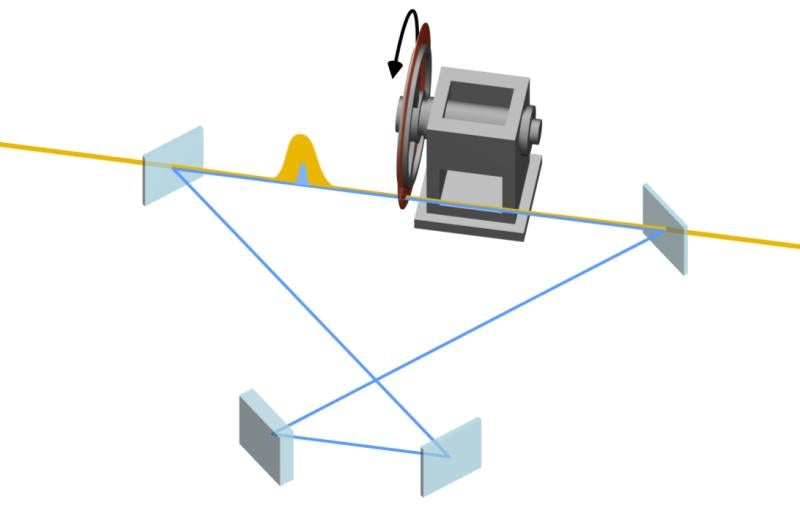
In 2018, researchers at the Department of Energy’s SLAC National Accelerator Laboratory developed a way to produce X-ray laser bursts which are several hundred attoseconds (or billionths of a billionth of a second) long. This method, called X-ray laser-enhanced attosecond pulse generation (XLEAP), allows scientists to investigate how electrons zipping around molecules jumpstart key processes in biology, chemistry, materials science and more.
“Electron motion is an important process by which nature can move energy around,” says SLAC scientist James Cryan. “A charge is created in one part of a molecule and it transfers to another part of the molecule, potentially kicking off a chemical reaction. It’s an important piece of the puzzle when you start to think about photovoltaic devices for artificial photosynthesis, or charge transfer inside a molecule.”
Now, researchers at SLAC’s Linac Coherent Light Source (LCLS) have rattled the electrons in a molecule with these attosecond pulses to create an excited quantum state and measure, in never-before-seen detail, how the electrons behave in this state. The results were published in Science earlier this month.
“XLEAP allows us to peer deep inside molecules and follow electron motion on its natural time scale,” says SLAC scientist Agostino Marinelli, who leads the XLEAP project. “This could provide insight into many important quantum mechanical phenomena, where electrons typically play a key role.”
Electronic messengers
Attosecond pulses are the shortest pulses generated at X-ray free-electron lasers like LCLS. The unique achievement of the XLEAP project has been to make attosecond pulses at the right wavelength to look inside the most important small atoms, such as carbon, nitrogen and oxygen. Like cameras with ultrafast shutter speeds, XLEAP pulses can capture the movements of electrons and other motions on an extremely fast timescale that could not be resolved before.
When X-ray pulses interact with matter, they can boost some of the most tightly bound core electrons in the sample to highly energetic states, known as core-excited states. Because they are so energetic, core-excited states are extremely unstable and will typically decay very quickly by releasing energy in the form of a fast electron, known as an Auger-Meitner electron. This phenomenon has historically been known as Auger decay but recently scientists have chosen to add the name of Lise Meitner, who first observed the phenomenon, in recognition of her broad-ranging contributions to modern atomic physics.
In their study, the researchers precisely tuned the wavelength of the X-rays from LCLS to create a quantum state of matter called a coherent superposition, a manifestation of the wavelike nature of matter. Similar to Schrödinger’s cat, which found itself both dead and alive at the same time, the excited electrons were simultaneously in different core-excited states. This meant they were orbiting the molecule along different trajectories at the same time.
To follow how this coherent superposition of core-excited states unfolded over time, the researchers created an ultrafast clock known as an ‘attoclock,’ where a rapidly rotating electric field from a circularly polarized laser pulse acts as the clock hand. The Auger-Meitner electrons released in the decay of the core-excited states were whirled around by the circularly polarized laser pulse before landing on the detector. The position in which an electron landed on the detector told the researchers the time at which it was ejected from the molecule. By measuring the ejection times of many Auger-Meitner electrons, the researchers were able to build up a picture of how the coherent superposition state was changing with a time resolution of just a few hundred attoseconds.
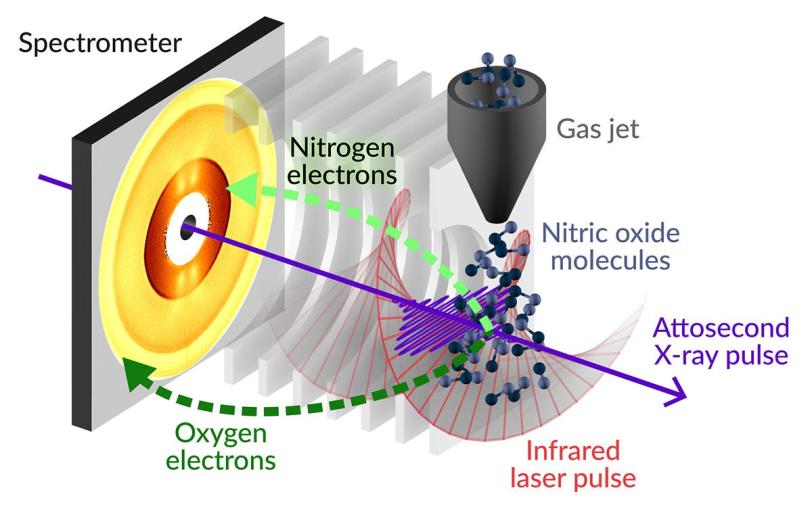
“It's the first time that we're able to track this particular phenomenon and directly measure the rate of electron emission,” says SLAC scientist and lead author Siqi Li. “Our technique takes us a step beyond just seeing the process happen and allows us to spy on the intricate electron behavior happening in the molecule within a few millionths of a billionth of a second. It gives us a really nice way to look inside the molecule and see what's happening on a very fast timescale.”
World-leading capability
To follow up on this experiment, the researchers are working on new measurements of more complex quantum behavior.
“In this experiment we are looking at the electronic behavior of a very simple model that you can almost solve with a pencil and paper,” says SLAC scientist and joint lead author Taran Driver. “Now that we’ve shown we can make these ultrafast measurements, the next step is to look at more complicated phenomena that theories are not yet able to accurately describe.”
The ability to make measurements on faster and faster timescales is exciting, Cryan says, because the first things that happen in a chemical reaction might hold the key to understanding what happens later.
“This research is the first time-resolved application of these ultrashort X-ray pulses, bringing us one step closer to doing really cool things like watching quantum phenomena evolve in real time,” he says. “It has the promise to become a world-leading capability that many people will be interested in for years to come.”
LCLS is a DOE Office of Science user facility. This research is part of a collaboration between researchers from SLAC, Stanford University, Imperial College London and other institutions. It was supported by the Office of Science.
Citation: S. Li, T. Driver et al. Science, 6 January 2022 (DOI: 10.1126/science.abj2096)
For questions or comments, contact the SLAC Office of Communications at communications@slac.stanford.edu.
SLAC is a vibrant multiprogram laboratory that explores how the universe works at the biggest, smallest and fastest scales and invents powerful tools used by scientists around the globe. With research spanning particle physics, astrophysics and cosmology, materials, chemistry, bio- and energy sciences and scientific computing, we help solve real-world problems and advance the interests of the nation.
SLAC is operated by Stanford University for the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time.