In brief: Breaking up buckyballs is hard to do
A new study shows how soccer ball-shaped molecules burst more slowly than expected when blasted with an X-ray laser beam.
As reported in Nature Physics, an international research team observed how soccer ball-shaped molecules made of carbon atoms burst in the beam of an X-ray laser. The molecules, called buckminsterfullerenes – buckyballs for short – consist of 60 carbon atoms arranged in alternating pentagons and hexagons like the leather coat of a soccer ball. These molecules were expected to break into fragments after being bombarded with photons, but the researchers watched in real time as buckyballs resisted the attack and delayed their break-up.
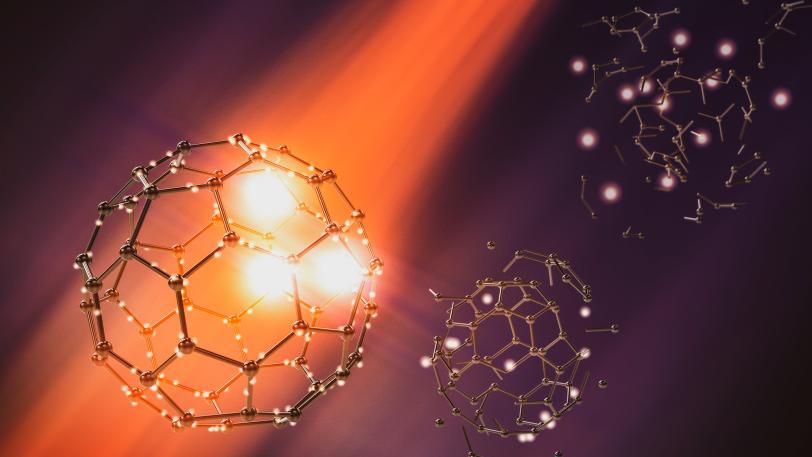
The team was led by Nora Berrah, a professor at the University of Connecticut, and included researchers from the Department of Energy’s SLAC National Accelerator Laboratory and the Deutsches Elektronen-Synchrotron (DESY) in Germany. The researchers focused their attention on examining the role of chemical effects, such as chemical bonds and charge transfer, on the buckyball’s fragmentation.
Using X-ray laser pulses from SLAC’s Linac Coherent Light Source (LCLS), the team showed how the bursting process, which takes only a few hundred femtoseconds, or millionths of a billionth of a second, unfolds over time. The results will be important for the analysis of sensitive proteins and other biomolecules, which are also frequently studied using bright X-ray laser flashes, and they also strengthen confidence in protein analysis with X-ray free-electron lasers (XFELs).
“This investigation uncovered for the first time the persistence of the molecular structure, which thwarted fragmentation over a timescale of hundreds of femtoseconds” Berrah says. “With the dawn of several new XFELs in the world, the findings lay the foundation for a deeper understanding of XFEL-induced radiation damage, which will have a strong impact on biomolecular imaging.”
The study also involved researchers from Imperial College London; University of Gothenburg in Sweden; University of Texas; Synchrotron SOLEIL in France; Kansas State University; Tohoku University in Japan; State University of New York at Potsdam; and Max Planck Institute for Nuclear Physics, Max Born Institute and University of Hamburg, all in Germany.
LCLS is a DOE Office of Science user facility. This research was funded by DOE’s Office of Basic Energy Sciences.
This article is based on press releases from DESY and UConn.
Citation
N. Berrah et al., Nature Physics, 23 September 2019 (10.1038/s41567-019-0665-7)
Contact
For questions or comments, contact the SLAC Office of Communications at communications@slac.stanford.edu.
SLAC is a vibrant multiprogram laboratory that explores how the universe works at the biggest, smallest and fastest scales and invents powerful tools used by scientists around the globe. With research spanning particle physics, astrophysics and cosmology, materials, chemistry, bio- and energy sciences and scientific computing, we help solve real-world problems and advance the interests of the nation.
SLAC is operated by Stanford University for the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit energy.gov/science.





