Solving the mysteries of a Trojan-horse mosquito toxin
An LCLS imaging technique reveals how a mosquito-borne bacterium deploys a toxin to kill mosquito larvae. Scientists hope to harness it to fight disease.
By Caitlyn Buongiorno
Mosquitoes carry some of the most well-known diseases plaguing the world, including malaria, Zika fever and West Nile virus. These mosquito-borne illnesses, while harmless to the mosquito, contribute to over one million deaths around the world every year.
One way of curbing these infection rates is to reduce the overall number of mosquitoes in an area that can bite people. The problem is that many of the mosquitocides used today are human-made chemical compounds that can have adverse effects on people and other animal populations, such as bees, crustaceans and fish.
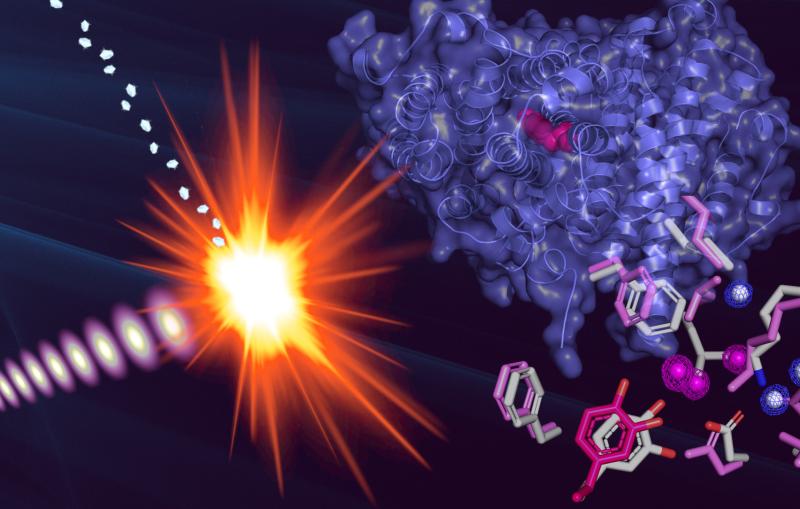
Now at the Department of Energy’s SLAC National Accelerator Laboratory, an international team led by Jacques-Philippe Colletier from University Grenoble Alpes in France has uncovered how bacteria use a Trojan-horse-like crystal to kill mosquitoes they infect. Researchers hope to harness it to kill mosquitoes that spread disease in humans.
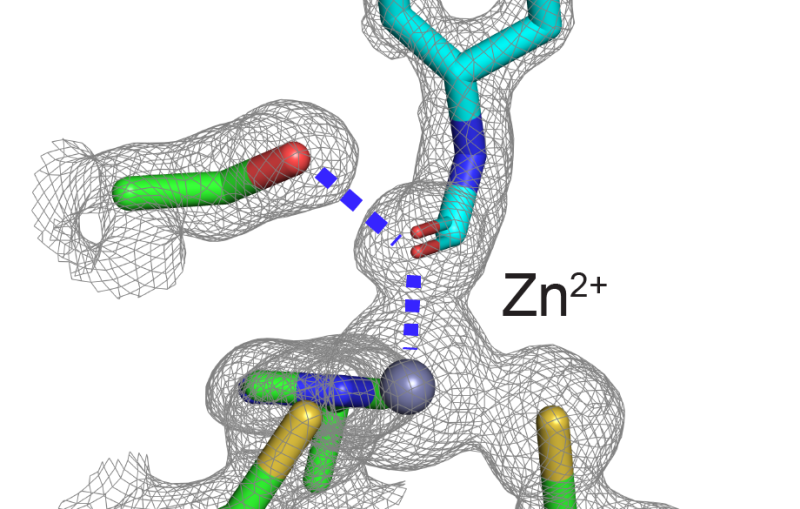
Cyt1Aa is one of the four protoxins – insecticidal crystal proteins that remain inactive until ingested by insect larvae – this bacterium produces to destroy mosquito larvae. With SLAC’s Linac Coherent Light Source (LCLS) X-ray laser, researchers were able to observe precisely how this crystal attacks the larvae. Their results were published in Nature Communications earlier this month.
“This is something that could not have been done before the advent of big X-ray lasers like LCLS,” says Raymond Sierra, an SLAC instrument scientist and co-author of the report. “For scientists sometimes seeing is believing and actually seeing these structures, from crystals thousands of times smaller than a human hair, and how they are behaving, could not have been done 10 years ago.”
To obtain a crystal structure like Cyt1Aa, researchers traditionally have directed an X-ray beam at the crystal and then interpreted the way the light diffracts into a detector to piece together the crystal’s atomic structure. The inherent problem with this technique is that the X-ray beams propagate damage throughout the sample. It becomes a race to collect data before the sample is destroyed.
With LCLS, researchers can hit a sample with an intense X-ray laser pulse that’s so short – just 40 femtoseconds, or millions of a billionth of a second, long – that they can collect the structural data they need in the instant before the sample is damaged.
“It’s analogous to lightning and thunder,” says Sierra. “If the X-ray is the lightning and the damage is the thunder, then you can get the information before the damage has a chance to propagate.”

Harvesting the crystals from the bacterium, the group used this technique to capture static images of the crystal at different pH levels to see subtle difference in the shapes of the pre-toxic and toxic forms. From this they were able to confirm just how much of a Trojan horse Cyt1Aa is and finally piece together how it takes down a cell once inside.
Before ingestion by a mosquito larva, Cyt1Aa protoxin is perfectly benign, the toxin hidden safely within the crystal’s structure. Once inside the larva, however, the crystal begins to unfold, exposing the toxin which then attaches itself to a cell wall.
With this study, scientists were able to finally determine the way the toxin breaks down the cellular wall of the mosquito larva’s gut, leading to its death. Multiple copies of the toxin attach themselves to the cell wall and, like blowing a hole in the side of a ship, each toxin creates a pore in the cell wall ripping the membrane apart. This pore causes the cell to lose its chemical equilibrium with the outside world, and it dies.
“It’s not very common for biological objects to self-organize into crystals,” says Sierra. “It’s very unique that the bacteria evolved to use this unorthodox form to effectively Trojan horse attack the larvae and do these nasty things.”
For starters, the crystal only unfolds at a very specific pH level that exists in the mosquito larvae gut, but not in humans. So a mosquitocide developed from this crystal would be more specifically targeted toward mosquitoes than the chemicals being used today.
While the study’s goal was to discover exactly how the toxin attacks mosquito larvae, researchers believe the crystal could be a potential mosquitocide for a number of reasons.
“There are paths that could take the information found in this study to make a more potent mosquitocide,” explains Mark Hunter, co-author and LCLS instrument scientist. “When you understand the structure of a protein, it can give you insight on an atomic level.” He adds that this could theoretically allow scientists to slightly alter the crystal to boost its performance or even target specific species of mosquito.
LCLS is a DOE Office of Science user facility. This work was partially supported by the DOE Office of Science and the National Institutes of Health.
Citation: G. Tetreau et al., Nature Communications, 2 March 2020 (10.1038/s41467-020-14894-w)
Contact
For questions or comments, contact the SLAC Office of Communications at communications@slac.stanford.edu.
SLAC is a vibrant multiprogram laboratory that explores how the universe works at the biggest, smallest and fastest scales and invents powerful tools used by scientists around the globe. With research spanning particle physics, astrophysics and cosmology, materials, chemistry, bio- and energy sciences and scientific computing, we help solve real-world problems and advance the interests of the nation.
SLAC is operated by Stanford University for the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time.