To create better batteries, SLAC researchers introduce disorder into the design
By instigating atomic disorder in lithium-ion battery materials, researchers created more stable materials that don’t expand, contract and degrade like traditional materials do.
By Emily Ayshford
Key takeaways:
- At the SLAC-Stanford Battery Center, researchers are redesigning batteries, starting at the atomic level.
- By introducing atomic disorder into lithium-ion battery materials, a team created batteries that last longer than traditional lithium-ion batteries.
- Designed with industry-standard materials and techniques, the technology is ready for rapid, large-scale adoption by battery manufacturers.
From data centers to the electrical grid, the demand for better, longer-lasting batteries is greater than ever before.
Today’s battery technology, however, is plagued by degradation. In lithium-ion batteries, for example, the materials inside expand and contract during each cycle, ultimately breaking down and limiting the life of the battery.
At the SLAC-Stanford Battery Center, a partnership between Stanford University’s Precourt Institute for Energy and the Department of Energy’s SLAC National Accelerator Laboratory, researchers are working to change that at the atomic level.
A research team found that by introducing a controlled amount of partial disorder into lithium-ion battery materials, they could create stable battery materials that don’t expand or contract.
The team published work in Nature Energy that showed these batteries have the potential to last longer than those currently used today.
The findings are part of the center’s broader effort to develop new, atomic-level design rules for next-generation energy storage.
“Our design is key to eliminating the expansion and contraction that plagues lithium-ion batteries,” said Junghwa Lee, a former postdoctoral researcher at Stanford University and SLAC National Accelerator Laboratory who is joint first author of the study with Zhelong Jiang, an associate scientist at SLAC. “We hope that this design is the basis for a new paradigm in batteries.”
Pillar-supported layering: a new design of stable cathode structure
Battery architecture generally involves two electrodes: the anode, which holds the ions as the battery is charged, and the cathode, into which the ions are released as the battery is discharged. An electrolyte solution shuttles the ions back and forth during charge and discharge.

The cathode’s composition mainly determines the battery’s energy density. One common type of cathode design uses layered oxide materials, which are made from alternating layers of lithium and transition metals, with oxygen ions gluing them together.
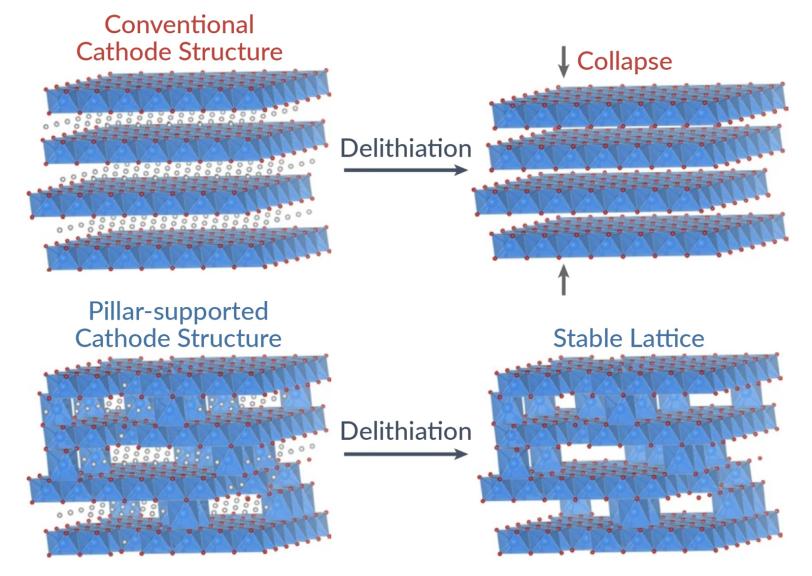
While effective for energy storage, these layers tend to shrink and expand when the battery is charged and discharged. This causes the battery to degrade over time and eventually fail.
“This layered oxide cathode is widely used, so this inherent structural instability is a major problem for high energy density and long-duration performance of batteries,” said William Chueh, director of the Stanford Precourt Institute for Energy, director of the SLAC-Stanford Battery Center, and a professor of materials science and engineering and of energy science and engineering at Stanford University who led the research.
SLAC-Stanford Battery Center
The SLAC-Stanford Battery Center enables translational research in electrochemical science and technology, bridging across fundamental science to deployment.

To get around this shrinkage, Chueh and his team started at the ground level. They didn’t want to rely on new materials and complex compositional modifications that would be difficult to integrate into the current battery manufacturing process.
Instead, they set their sights on a widely used cathode material – a nickel layered oxide – and changed the way the atoms are layered at the atomic level. Researchers already knew that removing lithium from the cathode as the battery is charged causes the structure to contract. Inserting non-lithium atoms into the layer is one strategy to prevent the collapse.
“However, instead of using foreign atoms to prevent the lattice from collapsing, we used the existing nickel atoms themselves,” Lee said.
The team released a small amount of oxygen as the battery was charged for the first time, causing some of the nickel to migrate between the atomic layers. These nickel atoms formed “pillars” that provide additional structural support to the layered cathode structure.
“When oxygen is released, the atoms in the materials start to move around,” Jiang said. “Researchers have previously used this technique to increase the energy density of the battery, but we used it as a way to permanently reorganize the atoms in the cathode structure.”
The result of this disorder is remarkable stability: The new lattice cathode structure exhibited near-zero strain, with less than 1% shrinkage when the battery was charged and discharged.
To verify these results, the team used the Stanford Synchrotron Radiation Lightsource (SSRL) at SLAC, utilizing extremely bright X-rays to study materials at the atomic and molecular levels, which provided direct evidence for the designed release of the oxygen and the resulting stabilization of the material.
A scalable solution for the market
This stable cathode material structure is achieved through a simple electrochemical activation step during the first cycle, making it highly suited for manufacturing, Lee said, because “every step involves processes that are already the industry standard, so it has the potential to be scaled up.”
Such an approach could also be used in other electrochemistry-centered energy storage systems. “Our design strategy is very generalizable,” Jiang said.
Director of the SLAC-Stanford Battery CenterWith even better design, we can continue to improve performance and reliability of batteries and make that technology suitable for the market.
Moving forward, the team plans to further optimize the design, increasing the cathode’s power density – how fast it can release energy – while testing other potential materials. As part of the larger mission of the SLAC-Stanford Battery Center, the goal is to harness the strengths of both SLAC and Stanford to increase the abundance of energy sources and improve energy resilience.
“With even better design, we can continue to improve performance and reliability of batteries and make that technology suitable for the market,” Chueh said.
Other institutions involved in the research include Korea Institute of Science and Technology and Kyung Hee University, Republic of Korea; University of California, Santa Barbara; Argonne National Laboratory; and BASF SE, Germany. Large parts of this work were supported by the California Research Alliance and the Department of Energy’s Office of Critical Minerals and Energy Innovation. SSRL is a DOE Office of Science user facility.
Citation: J. Lee et al., Nature Energy, 27 November 2025 (10.1038/s41560-025-01910-w)
For media inquiries, please contact media@slac.stanford.edu. For other questions or comments, contact SLAC Strategic Communications & External Affairs at communications@slac.stanford.edu.
About SLAC
SLAC National Accelerator Laboratory explores how the universe works at the biggest, smallest and fastest scales and invents powerful tools used by researchers around the globe. As world leaders in ultrafast science and bold explorers of the physics of the universe, we forge new ground in understanding our origins and building a healthier and more sustainable future. Our discovery and innovation help develop new materials and chemical processes and open unprecedented views of the cosmos and life’s most delicate machinery. Building on more than 60 years of visionary research, we help shape the future by advancing areas such as quantum technology, scientific computing and the development of next-generation accelerators.
SLAC is operated by Stanford University for the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time.





