Shapeshifting proteins provide electrons safe passage during photosynthesis
The results of a new scientific study reveal how photosynthetic reaction centers adapt to make light-driven reactions more efficient.
During photosynthesis, protein complexes within plants, algae, cyanobacteria and bacteria harvest energy from sunlight by shuttling electrons through a chain of molecules. Despite their important role in fueling life on Earth, how these photosynthetic reaction centers sustain the electron transfer process is not fully understood.
“If you think about the origin of life, people are quite convinced that the contrast between electron-rich chemical species coming from volcanoes and electron-poor chemical species coming from the ocean caused a cascade of electrons that started things off,” says University of Gothenberg scientist Richard Neutze. “And this cascade of electrons is happening in all biology all the time: When you eat food, you are taking advantage of electron movements to harvest the energy.”
Now, a team that includes researchers from the Department of Energy’s SLAC National Accelerator Laboratory has observed what happens in the reaction center of a bacterium in the trillionths of a second after it absorbs light. The results of their work, which reveal how proteins change shape to adjust to the movements of electrons, were published on Wednesday in Nature.
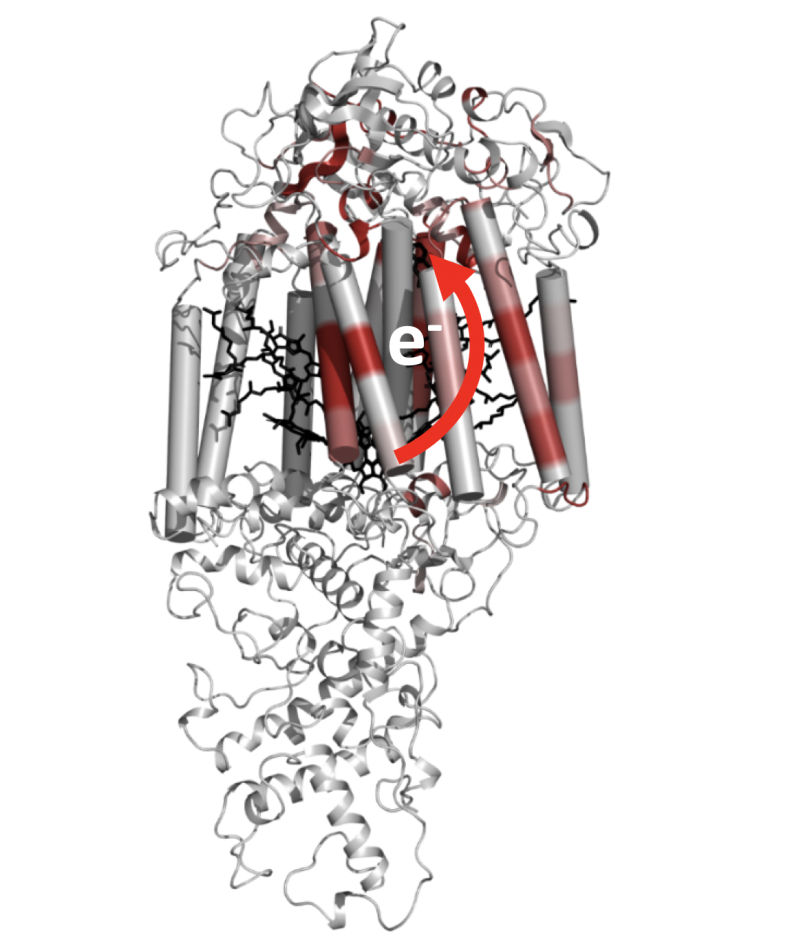
“This research shows us something quite fundamental about how proteins adapt to electron transfer to make these reactions more successful,” says Neutze, who led the study. “It provides new insight into how evolution has optimized the light-driven movements of charges in photosynthesis to achieve almost perfect overall efficiency.”
After kicking off the photosynthetic reaction with an optical laser pulse, the researchers studied the bacterium with X-rays from SLAC’s Linac Coherent Light Source (LCLS) to watch how the process unfolds. First, a pair of chlorophyll molecules within the reaction center absorbs the light, exciting electrons. The excited electrons then begin their journey to the acceptor molecule on the opposite side of the reaction center, resulting in what’s known as charge separation – the formation of a positive charge on the initial molecule and a negative charge on the acceptor. The team saw that as the electrons kicked into motion, the reaction center shifted its shape to make sure that the energy was used efficiently during these steps of the electron transfer reaction.
To follow up on these results, the researchers are investigating if they can watch the reaction unfold at even faster timescales and follow how the electrons move in greater detail.
SLAC scientist and co-author Sebastien Boutet says, “This research is an important step toward understanding the fundamental processes behind the ultrafast light-driven reactions that support life.”
LCLS is a DOE Office of Science user facility. This research was funded in part by the National Institutes of Health; the European Commission Marie Curie Training Networks; the European Union’s Horizon 2020 research and innovation program; the Swedish Research Council; the Swedish Foundation for Strategic Research; the Knut and Alice Wallenberg Foundation; the Academy of Finland; the BioExcel CoE project funded by the European Union; and the CSC-IT center in Espoo, Finland.
Citation: R. Dods et al., Nature, 2 December 2020 (10.1038/s41586-020-3000-7)
Contact
For questions or comments, contact the SLAC Office of Communications at communications@slac.stanford.edu.
SLAC is a vibrant multiprogram laboratory that explores how the universe works at the biggest, smallest and fastest scales and invents powerful tools used by scientists around the globe. With research spanning particle physics, astrophysics and cosmology, materials, chemistry, bio- and energy sciences and scientific computing, we help solve real-world problems and advance the interests of the nation.
SLAC is operated by Stanford University for the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time.





