Scientists watch gas-phase ions form and transform in real time
The results could lead to a better understanding of reactions with vital roles in chemistry and biology.
Ions are everywhere, from our daily surroundings to the cosmic expanse. Common table salt dissolves into sodium and chloride ions, regulating nerve impulses and muscle movements when absorbed by the body. In the sun, plasma, a gathering of ions in the gaseous state, undergoes nuclear fusion reactions, transmitting light and energy to Earth. One of the most common ions in everyday life is the lithium ion in batteries, powering devices like smartphones and electric cars. Ions play pivotal roles in various aspects of our lives, and understanding their processes and dynamics is crucial for science and technology.
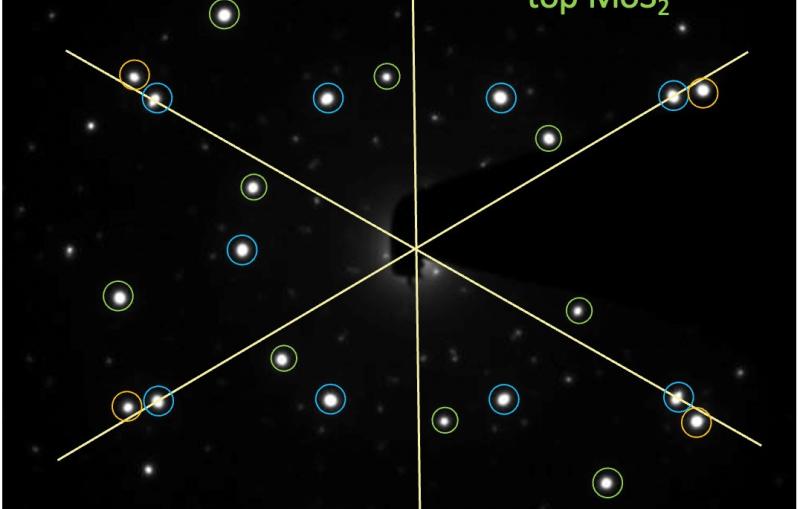
Until recently, capturing the moments of ion formation and transformation, especially in the gas phase, has been challenging. Now, scientists at the Department of Energy’s SLAC National Accelerator Laboratory and the Institute for Basic Science (IBS) in Korea, have demonstrated a way to follow ionization and structural changes in gas-phase molecules as they occur. The team used MeV-UED, an ultrafast “electron camera” at the Department of Energy’s SLAC National Accelerator Laboratory, which allowed them to observe ultrafast and ultrasmall movements of ion molecules. The results, published last week in Nature, could lead to a better understanding of reactions with vital roles in chemistry and biology.

This research team, known for previous breakthroughs in molecular dynamics, observed the formation and transformation of gas-phase ions. They looked closely at tiny charged particles called cations from a chemical named 1,3-dibromopropane (DBP) and discovered a fascinating phenomenon. These tiny particles stayed in a special, stable form called the 'dark state' for a very short time, about 3.6 trillionths of a second, before changing into a different shape that looks like a ring. Eventually, one of the bromine atoms (which is part of the DBP chemical) slowly broke off, leaving behind a special type of ion with a ring made of three atoms.
This study provides new insights into the intricate features and behavior of ions, which could lead to substantial contributions to future research endeavors in related fields such as astrochemistry and materials science.
"Despite the remarkable strides in science and technology, numerous captivating mysteries remain in the material world,” said co-author Hyotcherl Ihee, a scientist at the Institute for Basic Science. “This research, though unveiling just one more enigma of ions previously undiscovered, underscores the profound secrets awaiting our exploration."
MeV-UED is an instrument of SLAC’s Linac Coherent Light Source (LCLS) X-ray laser facility. LCLS is a DOE Office of Science user facility. This research was supported in part by the Office of Science (BES).
This article was adapted from a news release from the Institute for Basic Science.
Citation: J. Heo et al., Nature, 10 January 2024 (doi.org/10.1038/s41586-023-06909-5)
Contact
For questions or comments, contact the SLAC Office of Communications at communications@slac.stanford.edu.
About SLAC
SLAC National Accelerator Laboratory explores how the universe works at the biggest, smallest and fastest scales and invents powerful tools used by researchers around the globe. As world leaders in ultrafast science and bold explorers of the physics of the universe, we forge new ground in understanding our origins and building a healthier and more sustainable future. Our discovery and innovation help develop new materials and chemical processes and open unprecedented views of the cosmos and life’s most delicate machinery. Building on more than 60 years of visionary research, we help shape the future by advancing areas such as quantum technology, scientific computing and the development of next-generation accelerators.
SLAC is operated by Stanford University for the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time.