SLAC Experiment Finds Key to Natural Detoxifier’s Reactivity
Results Have Implications for Health, Drug Design and Chemical Synthesis
Researchers working at the Department of Energy’s SLAC National Accelerator Laboratory have discovered that a mere 9-trillionths-of-a-meter reduction in the length of a chemical bond dramatically boosts the reactivity of a family of molecules that helps keep humans and many other organisms healthy.
This result, reported recently in Nature Chemistry, is expected to help scientists design and manufacture new and more effective medicines.
The molecules, called cytochrome P450s, are known as “nature’s detoxifiers,” said Courtney Krest Roach, a beamline scientist at SLAC’s Stanford Synchrotron Radiation Lightsource (SSRL) – a DOE Office of Science User Facility – and first author of the paper.
Since cytochrome P450 was discovered more than 50 years ago, more than 11,000 variants have been identified in every kingdom of life, including animals, plants, fungi and bacteria. Some 57 different P450s are found in humans, typically in the liver, where they help break down poisons and unwanted molecules. They are also essential in the body’s manufacture of certain hormones, and chemists hope to be able to use them in the future to catalyze the creation of complex molecules for use in medicine and industry.
“Our bodies use P450s to perform one step in the metabolism of some 75 percent of pharmaceuticals,” Roach said. “And the P450s’ extreme selectivity can enable much more direct and efficient synthesis of a number of high-value hydrocarbons and organic molecules.”
Molecular Structure is Key
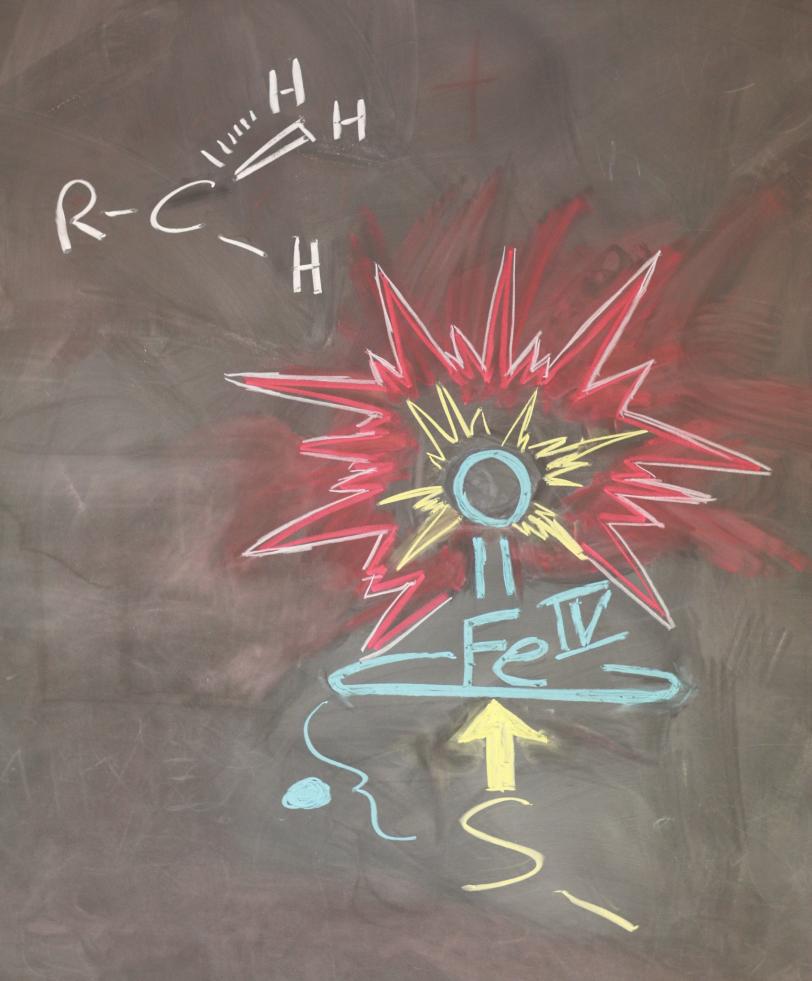
Each cytochrome P450 is optimized to catalyze an initial “activation” step in a specific chemical reaction. They all share the same basic atomic structure: A central iron atom is surrounded by four nitrogen atoms, with a sulfur-based structure sticking out from one side of the ring. Activation creates an intermediate compound that has an oxygen atom double-bonded to the iron atom; it sticks out from the other side of the ring.
Because the P450s’ intermediate compounds were short-lived, chemists who were trying to learn how they worked tried studying a slightly simpler molecule, called CPO, which had a more stable intermediate. But they found it was much less reactive than the P450s – in some cases only one-fortieth as reactive. Researchers needed to know why, because a more reactive P450 can break stronger bonds, metabolize new medicines or make different final products.
The new experiments at SSRL were led by Penn State University Professor Michael Green, who was Roach’s PhD advisor. The research team used the absorption of X-rays by the intermediate forms of CPO and P450s to make ultraprecise measurements of their atomic structures. The results showed that the big difference in reactivity was caused by a miniscule, 9-trillionths-of-a-meter difference in the lengths of their iron-sulfur bonds.
Small Changes, Big Impact
Slight changes in hydrogen bonding near the central iron atom shortened the length of the P450s’ iron-sulfur bond by 4 percent. This changed the distribution of nearby electrons in a way that weakened and lengthened the iron-oxygen double bond on the opposite side of the ring. This, in turn, made it easier for the oxygen atom to carry out its part in the activation process, thus increasing P450s’ reactivity.
“SSRL is the only place that we could do these experiments with the necessary quality,” Roach said.
In their “News & Views” commentary on this research published in the same issue of Nature Chemistry, University of Illinois professors Ilia Denisove and Stephen Silgar said this result is a significant breakthrough that will provide the basis for researchers to design and make new catalysts and adapt P450-based systems for use in biotechnology.
SSRL's Structural Molecular Biology program is supported by the National Institutes of Health and the DOE’s Office of Biological and Environmental Research.
Citation: C. Krest et al., Nature Chemistry, 3 August 2015 (10.1038/nchem.2306)
For questions or comments, contact the SLAC Office of Communications at communications@slac.stanford.edu.
SLAC is a multi-program laboratory exploring frontier questions in photon science, astrophysics, particle physics and accelerator research. Located in Menlo Park, Calif., SLAC is operated by Stanford University for the U.S. Department of Energy's Office of Science.
SLAC National Accelerator Laboratory is supported by the Office of Science of the U.S. Department of Energy. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.