In a leap for battery research, machine learning gets scientific smarts
The latest advance from a research collaboration with industry could dramatically accelerate the development of sturdier batteries for fast-charging electric vehicles.
By Glennda Chui
Scientists have taken a major step forward in harnessing machine learning to accelerate the design for better batteries: Instead of using it just to speed up scientific analysis by looking for patterns in data, as researchers generally do, they combined it with knowledge gained from experiments and equations guided by physics to discover and explain a process that shortens the lifetimes of fast-charging lithium-ion batteries.
It was the first time this approach, known as “scientific machine learning,” has been applied to battery cycling, said Will Chueh, an associate professor at Stanford University and investigator with the Department of Energy’s SLAC National Accelerator Laboratory who led the study. He said the results overturn long-held assumptions about how lithium-ion batteries charge and discharge and give researchers a new set of rules for engineering longer-lasting batteries.
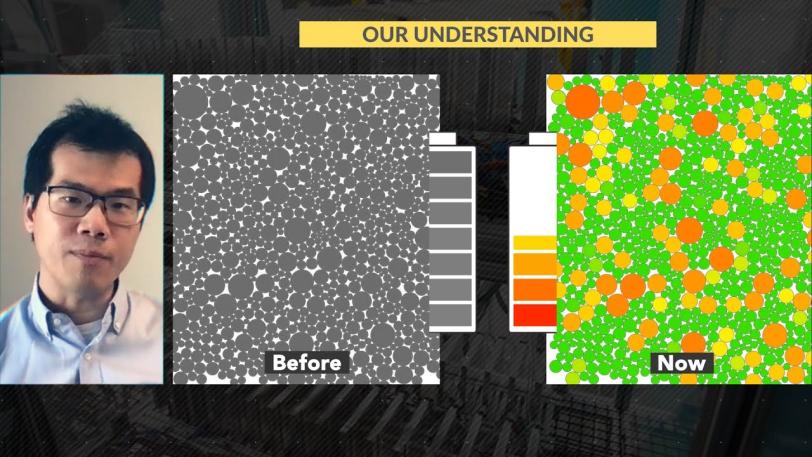
Battery Dynamics / Fast-charging / Major Discovery
Olivier Bonin/SLAC National Accelerator Laboratory
The research, reported today in Nature Materials, is the latest result from a collaboration between Stanford, SLAC, the Massachusetts Institute of Technology and Toyota Research Institute (TRI). The goal is to bring together foundational research and industry know-how to develop a long-lived electric vehicle battery that can be charged in 10 minutes.
"Battery technology is important for any type of electric powertrain," said Patrick Herring, senior research scientist for Toyota Research Institute. “By understanding the fundamental reactions that occur within the battery we can extend its life, enable faster charging and ultimately design better battery materials. We look forward to building on this work through future experiments to achieve lower-cost, better-performing batteries."

A trio of advances
The new study builds on two previous advances where the group used more conventional forms of machine learning to dramatically accelerate both battery testing and the process of winnowing down many possible charging methods to find the ones that work best. While these studies allowed researchers to make much faster progress – reducing the time needed to determine battery lifetimes by 98%, for instance – they didn’t reveal the underlying physics or chemistry that made some batteries last longer than others, as the latest study did.
Combining all three approaches could potentially slash the time needed to bring a new battery technology from the lab bench to the consumer by as much as two-thirds, Chueh said.
“In this case, we are teaching the machine how to learn the physics of a new type of failure mechanism that could help us design better and safer fast-charging batteries,” Chueh said. “Fast charging is incredibly stressful and damaging to batteries, and solving this problem is key to expanding the nation’s fleet of electric vehicles as part of the overall strategy for fighting climate change.”
The new combined approach can also be applied to developing the grid-scale battery systems needed for a greater deployment of wind and solar electricity, which will become even more urgent as the nation pursues recently announced Biden Administration goals of eliminating fossil fuels from electric power generation by 2035 and achieving net-zero carbon emissions by 2050.

Zooming in for closeups
The new study zoomed in on battery electrodes, which are made of nano-sized grains glommed together into particles. Lithium ions slosh back and forth between the cathode and anode during charging and discharging, seeping into the particles and flowing back out again. This constant back-and-forth makes particles swell, shrink and crack, gradually decreasing their ability to store charge, and fast charging just makes things worse.
To look at this process in more detail, the team observed the behavior of cathode particles made of nickel, manganese and cobalt, a combination known as NMC that’s one of the most widely used materials in electric vehicle batteries. These particles absorb lithium ions when the battery discharges and release them when it charges.
Stanford postdoctoral researchers Stephen Dongmin Kang and Jungjin Park used X-rays from SLAC’s Stanford Synchrotron Radiation Lightsource to get an overall look at particles that were undergoing fast charging. Then they took particles to Lawrence Berkeley National Laboratory’s Advanced Light Source to be examined with scanning X-ray transmission microscopy, which homes in on individual particles.

The data from those experiments, along with information from mathematical models of fast charging and equations that describe the chemistry and physics of the process, were incorporated into scientific machine learning algorithms.
“Rather than having the computer directly figure out the model by simply feeding it data, as we did in the two previous studies, we taught the computer how to choose or learn the right equations, and thus the right physics,” said Kang, who performed the modeling with MIT graduate student Hongbo Zhao, working with chemical engineering professor Martin Bazant.
The rich-get-richer effect
Until now, scientists had assumed that the differences between particles were insignificant and that their ability to store and release ions was limited by how fast lithium could move inside the particles, Kang said. In this way of seeing things, lithium ions flow in and out of all the particles at the same time and at roughly the same speed.
But the new approach revealed that the particles themselves control how fast lithium ions move out of cathode particles when a battery charges, he said. Some particles immediately release a lot of their ions while others release very few or none at all. And the quick-to-release particles go on releasing ions at a faster rate than their neighbors – a positive feedback, or “rich get richer,” effect that had not been identified before.

“We now have a picture – literally a movie – of how lithium moves around inside the battery, and it’s very different than scientists and engineers thought it was,” Kang said. “This uneven charging and discharging puts more stress on the electrodes and decreases their working lifetimes. Understanding this process on a fundamental level is an important step toward solving the fast charging problem.”
The scientists say their new method has potential for improving the cost, storage capacity, durability and other important properties of batteries for a wide range of applications, from electric vehicles to laptops to large-scale storage of renewable energy on the grid.
“It was just two years ago that the 2019 Nobel Prize in chemistry was awarded for the development of rechargeable lithium-ion batteries, which dates back to the 1970s,” Chueh said. “So I am encouraged that there’s still so much to learn about how to make batteries better.”
This research was funded by Toyota Research Institute. The Stanford Synchrotron Radiation Lightsource and Advanced Light Source are DOE Office of Science user facilities, and work there was supported by the DOE Office of Science and the DOE Advanced Battery Materials Research Program.
Citation: Jungjin Park et al., Nature Materials, 8 March 2021 [10.1038/s41563-021-00936-1]
Press Office Contact: Manuel Gnida, mgnida@slac.stanford.edu, (650) 926-2632
SLAC is a vibrant multiprogram laboratory that explores how the universe works at the biggest, smallest and fastest scales and invents powerful tools used by scientists around the globe. With research spanning particle physics, astrophysics and cosmology, materials, chemistry, bio- and energy sciences and scientific computing, we help solve real-world problems and advance the interests of the nation.
SLAC is operated by Stanford University for the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time.