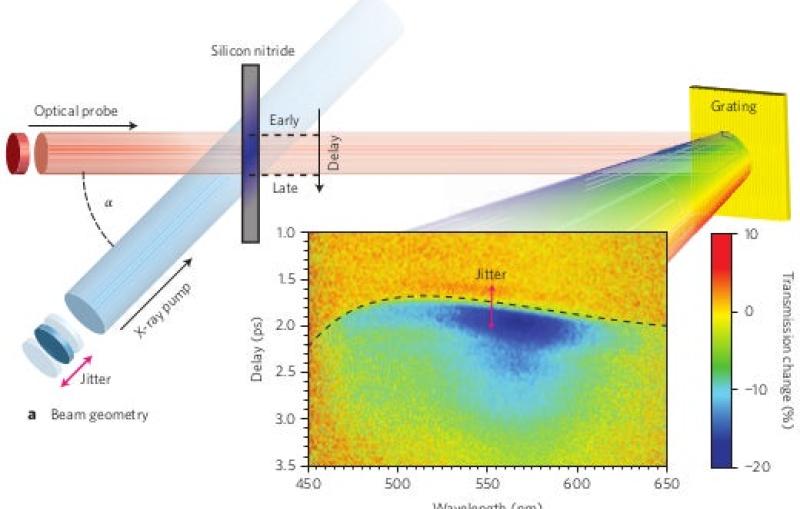
With up to a million X-ray flashes per second, 8,000 times more than its predecessor, it transforms the ability of scientists to explore atomic-scale, ultrafast phenomena that are key to a broad range of applications, from quantum materials to clean...