SLAC topics
Biological sciences
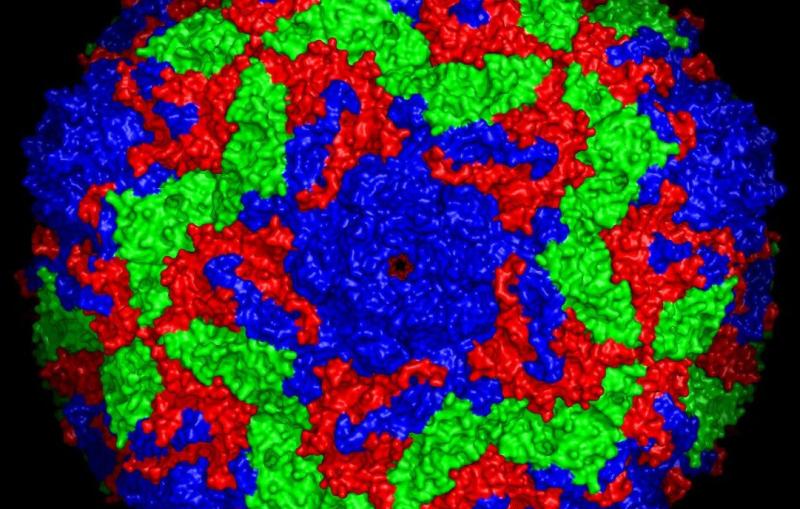
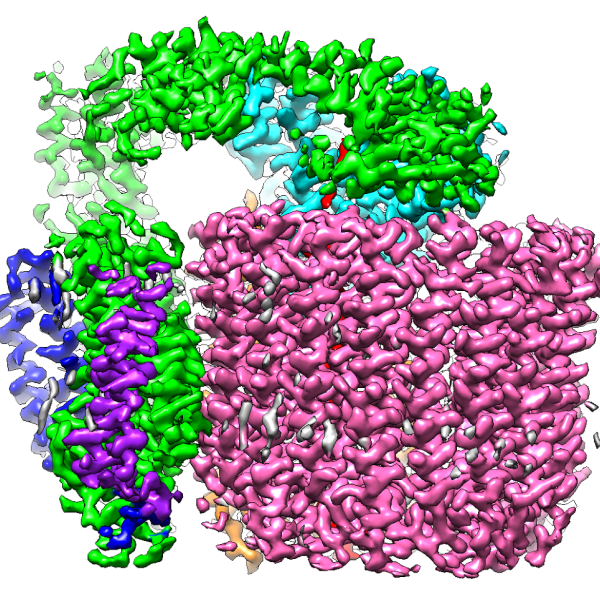
Tiny microbes and molecular machines have an outsized impact on human health, and they play key roles in the vast global cycles that shape climate and make carbon and nitrogen available to all living things.
Related link:
Science of life